Is there a domestic version of Trametinib and information on domestic generic drugs?
Trametinib (Trametinib) is a MEK inhibitor used to treat BRAF V600 mutation-related tumors. It is mainly used for targeted therapy in patients with melanoma and some non-small cell lung cancer. Currently, there is no domestic version in China, and patients can only be treated with imported original drugs. Trametinib is already on the market in China and has been included in the medical insurance system, greatly improving patient accessibility.
In the domestic market, the common specifications of trametinib include 0.5mg30 tablets and 2mg30 tablets. The latest selling price of 0.5mg30 tablets is around three to four thousand yuan, while the price of 2mg30 tablets is higher, with the latest selling price ranging from more than nine thousand to more than ten thousand yuan. For specific medical insurance reimbursement prices and usage policies, patients need to consult the local hospital pharmacy to obtain the most accurate information.
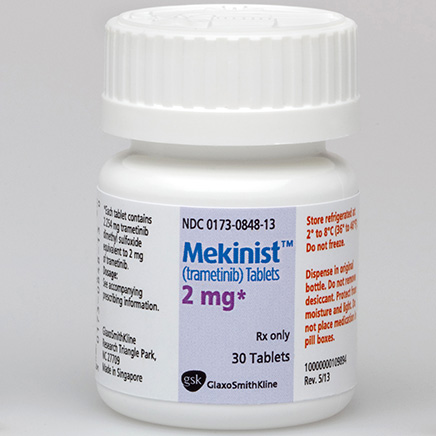
In foreign markets, the price of trametinib varies greatly. For example, the original version of trametinib 2mg*30 tablets in the Turkish market is priced at about 7,000 to 8,000 yuan. It is worth noting that the Ministry of Health of Laos has approved the marketing of generic trametinib drugs. The pharmaceutical ingredients of this type of generic drugs are basically the same as the original drugs. The latest price is more than 1,000 yuan, which is significantly lower than the original drug, providing an economically feasible option for overseas patients.
Overall, trametinib still relies on imported original drugs in China, but with medical insurance coverage and the price reference of overseas generic drugs, the financial burden on patients is expected to be gradually eased in the future. When choosing medication, patients should take into account their own condition, doctor's advice, and financial status, as well as drug efficacy, price, and accessibility to formulate the best treatment plan.
Reference materials:https://www.drugs.com/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















