Is Quizartinib available in the country?
As of 2025, Quizartinib (Quizartinib)’s original drug has not yet been officially launched for sale in mainland China, so it is temporarily unavailable for purchase in domestic pharmacies or hospitals. Its original research company is Japan's Daiichi Sankyo, with the trade name Vanflyta. It has been approved for marketing in the United States, Japan and the European Union for the treatment of FLT3-ITD-positive acute myeloid leukemia (AML). It is the world's first type II FLT3 inhibitor that specifically targets this mutation.
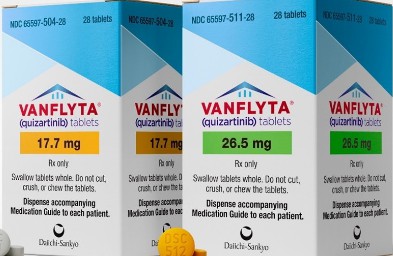
In overseas pharmacies and authorized channels, the common dosage forms of Quizartinib are17.7 mg (pack of 28 tablets) and 26.5 mg (pack of 56 tablets). The price per box is generally between RMB 80,000 and RMB 100,000, depending on the exchange rate, regional taxes and availability. Some patients can purchase European original drugs through overseas medical channels, but they must do so under the guidance of a qualified doctor's prescription to avoid the risk of illegal purchase. In addition, generic versions of drugs have also appeared in overseas markets. Among them, the well-known generic version of Quizartinib produced by Lucius Pharmaceuticals in Laos is packed with 14 tablets per box and sells for about RMB 1,000. The ingredients of the drug are consistent with the original drug. However, there is still a lack of sufficient verification in terms of bioequivalence and long-term safety data, so it is not recommended to purchase and use it by yourself.
China has not yet approved any generic drug of Quizartinib for marketing, and the drug has not yet been included in the medical insurance reimbursement list. However, some large domestic tumor centers and hematology institutions have launched clinical research projects related to Quizartinib, and some patients can obtain the drug through controlled clinical channels. With the advancement of the international reform of my country's drug registration system, it is expected that Quizartinib will enter the Chinese market in the next two to three years. For patients with FLT3 mutant AML, quizartinib represents a new direction in targeted precision therapy, and domestic clinical accessibility issues are gradually improving with the improvement of mechanisms such as accelerated policy approval and cross-border drug introduction.
Reference materials:https://go.drugbank.com/drugs/DB12874
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















