Brief description of the instructions for use of the Indian version of regorafenib
Regorafenib is a multi-target tyrosine kinase inhibitor that blocks tumor cell signaling by inhibiting kinase activity, inhibits proliferation, invasion and metastasis, and has anti-immunosuppressive effects. Clinically, it is mainly used to treat patients with metastatic colon cancer, gastrointestinal stromal tumors and liver cancer, and can prolong survival. However, the high price of the original drug has discouraged many patients. Indian regorafenib provides patients with a relatively economical treatment option. However, some patients are unclear about the instructions for its use, which will be covered in this article.
1. Basic information about drugs
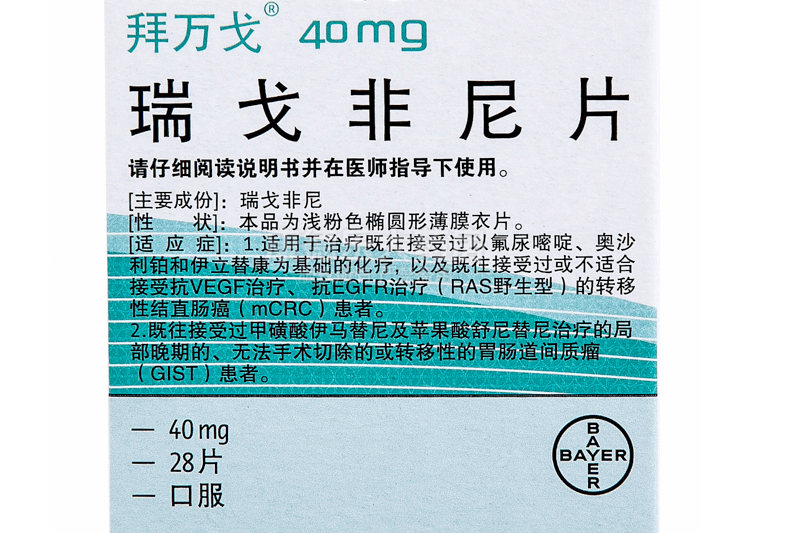
The Indian version of Regorafenib usually refers to the generic drug produced by NATCO Pharmaceuticals in India.The generic name is Regorafenib tablets (Regorafenib) and the trade name is Regonat, but the main ingredient is Regorafenib. The common specification of this product is 40mg*28 tablets, and the price is about more than 1,000.
2. Indications, usage and dosage
Regofenib is indicated for the treatment of a variety of advanced or metastatic cancers, including metastatic colorectal cancer (mCRC), gastrointestinal stromal tumor (GIST), and hepatocellular carcinoma (HCC). The recommended dose is 160mg (i.e. 4 tablets) per day, taken orally in the first 21 days of each course of treatment, and every 28 days constitutes a complete course of treatment. Patients should swallow the tablet whole with water at the same time every day, after a low-fat breakfast. If a dose is missed, it should not be taken on the same day.
3. Adverse reactions
The most common adverse reactions (≥30%) after taking the Indian version of regorafenib include fatigue/fatigue, decreased appetite, hand and foot skin reactions (such as palmar and plantar erythrodysesthesia), diarrhea, oral mucositis, weight loss, infection, hypertension and dysphonia, etc. Patients need to pay close attention to their symptoms. If they have severe or persistent adverse reactions, they should seek medical treatment promptly and may need to adjust the dosage or discontinue the medication.
4. Notes
1. Bleeding risk: For severe or life-threatening bleeding events, regorafenib therapy should be permanently discontinued.
2. Dermatologic Toxicity: Depending on the severity and persistence of dermatologic toxicity, interruption, dose reduction, or discontinuation of treatment may be necessary.
3. Hypertension management: Patients with severe or uncontrollable hypertension should temporarily or permanently terminate treatment and conduct blood pressure monitoring and adjustment under the guidance of a doctor.
4. Cardiac ischemia and infarction: For patients with new or acute cardiac ischemia/infarction, regorafenib should be withheld until the acute ischemic event is resolved.
5. Other serious reactions: such as reversible posterior leukoencephalopathy syndrome (RPLS), gastrointestinal perforation or fistula, wound healing complications, etc., all require immediate termination of treatment.
5. Storage conditions
The Indian version of regorafenib should be stored in a sealed container at room temperature and out of reach of children. Desiccant is included in the cap to prevent accidental ingestion. Please keep the bottle cap tightly closed to prevent the medicine from being exposed to heat and moisture.
In summary, the Indian version of regorafenib, as a multi-target small molecule inhibitor, has shown significant efficacy in the treatment of various advanced cancers. However, its use must strictly follow medical advice, pay close attention to adverse reactions, and make timely adjustments to the treatment plan according to the specific situation. Patients should maintain close communication with their doctors during medication to ensure safe and effective treatment.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















