The full version of the drug instructions for Dacomitinib/Dozerun
1. Common name and product name
Dacomitinib/Dacomitinib (Dacomitinib) is an oral small molecule targeted drug with trade names including Vizimpro and Dozerun. As a second-generation irreversible epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor, it plays a key role in the targeted treatment of advanced non-small cell lung cancer (NSCLC), especially for patients carrying EGFR sensitive mutations.
2. Indications
Dacomitinib is approved for the first-line treatment of patients with metastatic non-small cell lung cancer who have been found in molecular testingEGFR exon 19 deletions or exon 21 L858R substitution mutations. This indication is determined based on clinical data approved by the US FDA, providing a scientific basis for precise targeted therapy. By selecting patients who meet the mutation conditions to use dacomitinib, it can effectively inhibit tumor proliferation and improve the therapeutic effect.
3. Usage and dosage
Before using dacomitinib, doctors will evaluate the suitability based on the EGFR mutation type of the patient's tumor specimen and decide whether to perform first-line targeted therapy. The recommended dose is 45 mg orally once daily with or without food until disease progression or intolerable toxicity occurs.
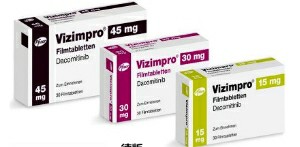
If the patient experiences obvious adverse reactions, the dose can be adjusted in stages: the first dose can be reduced to 30 mg per day If side effects still need to be controlled, the second dose can be reduced to 15 mg per day. In addition, drug interactions should be noted, especially proton pump inhibitors (PPIs) that may affect the absorption of dacomitinib. For patients who need to take H2 receptor antagonists, dacomitinib should be taken at least 6 hours before or at least 10 hours after taking H2 receptor antagonists to ensure drug efficacy. Topically acting antacids may be used as an alternative.
4. Adverse reactions
Common adverse reactions of dacomitinib in clinical applications include gastrointestinal and skin system reactions, among which diarrhea, rash, paronychia, stomatitis, dry skin and alopecia occur more frequently. In addition, some patients may experience symptoms such as weight loss, decreased appetite, cough, and itching. Abnormal laboratory indicators also require attention, including anemia, lymphopenia, hypoalbuminemia and blood electrolyte abnormalities, such as hypokalemia, hypocalcemia, hypomagnesemia, etc. Liver function indicators such as elevated ALT and AST, hyperbilirubinemia, elevated creatinine and alkaline phosphatase have also been reported from time to time.
5. Storage conditions
Dacomitinib should be stored20°C to 25°C (68°F to 77°F), with short-term excursions in the temperature range of 15°C to 30°C (59°F to 86°F). Medications must be kept sealed in their original packaging and protected from moisture and direct sunlight to ensure stable efficacy.
6. Mechanism of action
Dacomitinib blocks the key signaling pathways of cancer cells by irreversibly inhibiting the kinase activity of EGFR family members (EGFR/HER1, HER2 and HER4) and some EGFR activating mutations (exon 19 deletion or exon 21 L858R). Laboratory studies have shown that dacomitinib also inhibits the activities of kinases such as DDR1, EPHA6, LCK, DDR2 and MNK1 at clinically relevant concentrations. Animal model studies further demonstrate that dacomitinib inhibits EGFR and HER2 autophosphorylation in subcutaneously transplanted human tumor xenografts, thereby preventing tumor growth. In an intracerebral transplant model of oral administration, it also showed anti-tumor activity against EGFR amplification-driven tumors, suggesting its possible efficacy in patients with intracranial metastasis.
7. Clinical application tips
When treated with dacomitinib, patients should strictly follow the doctor's instructions and follow up regularly. Early identification of side effects, dose adjustment, and lifestyle intervention can maximize efficacy and maintain quality of life.
Reference materials:https://www.pfizer.com/products/product-detail/vizimpro
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















