Mitotane (mitotane) instructions
1.Indications and Usage
Mitotane is indicated for the treatment of patients with inoperable functional or non-functioning adrenocortical carcinoma.
2.Dose and Administration
1.Recommended dosage
The recommended initial dose of mitotane is 2 grams to 6 grams taken orally in three or four divided doses per day. Gradually increase the dose until the blood concentration reaches 14-20mg/L, or reaches the tolerance level.
Mitotane is a cytotoxic drug. Follow applicable special handling and disposal procedures.
2.Dose modification
Adrenal crisis in shock or severe trauma: Withhold mitotane until recovery.
Central Nervous System Toxicity: Discontinue Mitotane until symptoms resolve. 7to10days after symptoms disappear, restart at a lower dose (e.g., reduce 500-1000 mg).
Three. Dosage form and strength
500 white, round, biconvex, scored tablets, split in two, with "BL" printed on the other side instead of "L1" .
4.Contraindications
No.
Five. Warnings and Precautions
1.Adrenal crisis in shock or severe trauma
TakingmitotaneIn patients, adrenal crisis occurs in the setting of shock or severe trauma and the response to shock is impaired. Use hydrocortisone, monitor for worsening of shock symptoms, and discontinue mitotane until resolution occurs.
2.Central nervous system toxicity
CNS toxicity, including sedation, somnolence, and dizziness, may occur with mitotanetreatment. Mitotane plasma concentrations exceeding20 mcg/mLare associated with a higher incidence of toxicity.
3.Adrenal Insufficiency
Treatment withmitotanecan result in adrenal insufficiency. Steroid replacement therapy was performed as clinically indicated. Measure free cortisol and adrenocorticotropic hormone(ACTH)levels for optimal steroid replacement.
4.Embryo-Fetal Toxicity
Taking it by pregnant women Mitotane can cause harm to the fetus. Abnormal pregnancy outcomes, such as preterm birth and early pregnancy loss, may occur in patients exposed to mitotane during pregnancy. Inform pregnant women of potential risks to the fetus. As long as mitotane plasma levels are detectable, females of reproductive potential are advised to use effective contraception during treatment with mitotane and after discontinuation of treatment.
5.Large ovarian cysts in premenopausal women
Large ovarian cysts, usually bilateral and multiple, have been reported in premenopausal patients receivingmitotane. Complications of these cysts, including adnexal torsion and hemorrhagic cyst rupture, have been reported. In some cases, improvement occurred after mitotane was discontinued. Advise female patients to seek medical care if they experience gynecological symptoms such as vaginal bleeding and /or pelvic pain.
6. Adverse reactions
Adrenal crisis in the setting of shock or severe trauma, Central nervous system toxicity, Adrenal insufficiency, Large ovarian cyst.
Common adverse reactions during mitotane treatment include: Anorexia, nausea, vomiting and diarrhea(80%), Depression, dizziness or vertigo (15%-40%), Skin rash (15%), Neutropenia, Growth retardation , Hypothyroidism, Confusion, headache, ataxia, mental disorder, weakness, dysarthria, Macular hepatitis, elevated liver enzymes gynecomastia, Hypercholesterolemia, hypertriglyceridemia, Reduced blood androstenedione in women, Reduced blood testosterone,Sex hormone binding globulin increased in women and men, and blood free testosterone decreased in men.
Less common adverse reactions include:blurred vision, diplopia, lens opacification, retinopathy, prolonged bleeding time, hematuria, hemorrhagic cystitis, proteinuria, hypertension, orthostatic hypotension, flushing, generalized pain, pyrexia, and hypogonadism(Males)
7. Drug interactions
1.SomeCYP3Asubstrates
Mitotane is a strongCYP3A inducer. Concurrent use ofmitotanemay decreasethe concentrations of CYP3Asubstrates, thereby reducing the efficacy of these substrates. Avoid coadministration ofmitotanewith certainCYP3A4substrates as minimal concentration changes may result in treatment failure. If concomitant use cannot be avoided, increase the CYP3A substrate dose according to the approved product labeling.
2.Warfarin
When coumarin anticoagulants are administered to patients receivingmitotane, coagulation tests should be monitored and the anticoagulant dose adjusted as needed.
8. Use among specific groups of people
1.Pregnancy
Mitotanecan cause harm to the fetus. Limited postmarketing cases have been reported of preterm birth and early pregnancy loss in women treated with this product during pregnancy. Animal reproduction studies have not been conducted with mitotane. Inform pregnant women of potential risks to the fetus. The background risk of major birth defects and miscarriage in the populations shown is unknown. In the general U.S. population, the estimated background risks of major birth defects and miscarriage in clinically recognized pregnancies are 2-4% and 15-20% respectively.
2.Breastfeeding
Mitotan is excreted in human milk; however, mitotane's effects on breastfed infants or on milk production are not known. Because of the potential for serious adverse reactions in breastfed infants, lactating women are advised not to breastfeed during treatment with mitotane and after interruption of treatment as long as mitotane plasma levels are detectable.
3.Females and men of reproductive potential
Taking it by pregnant womenMitotancan cause harm to the fetus[See Use in Specific Populations. Advise female patients of reproductive potential to use effective contraception during treatment with mitotane and after interruption of treatment as long as mitotane plasma levels are detectable.
4.Pediatric use
Safety and effectiveness in pediatric patients have not been established.
5.Medication for the Elderly
Clinical studies of mitotane did not include sufficient numbers of patients 65 years and older to determine whether they responded differently than younger patients. Other reported clinical experience has found no difference in response between older and younger patients. In general, dosage selection in elderly patients should be cautious, often starting at the lower end of the dosage range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and concomitant disease or other medications.
6.Liver function impairment
Hepatic impairment may interfere with the metabolism of mitotane and accumulation of the drug may occur. Use mitotane with caution in patients with hepatic impairment.
9. Description
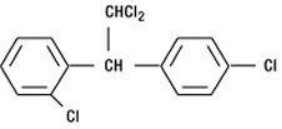
Mitotane is an oral adrenocytotoxic agent. The chemical name is ()-1, 1-dichloro-2-(o-chlorophenyl)-2-(p-chloro Phenyl)ethane(also known as o, p′-DDD). The chemical structure is:
xa0
Mitotan is a white granular solid composed of transparent colorless crystals. It is odorless and has a slightly pleasant aromatic smell. It is soluble in ethanol and has a molecular weight of 320.05.
The inactive ingredients in mitotane are:microcrystalline cellulose, polyethylene glycol3350, silicon dioxide, and starch.
10. Clinical Pharmacology
1.Mechanism of action
Mitotan is an adrenal cytotoxic agent with an unknown mechanism of action. Mitotane alters the peripheral metabolism of steroids and directly inhibits the adrenocortex.据报道,在皮质类固醇浓度没有降低的情况下,17-羟基皮质类固醇减少,而6-β-羟基皮质醇的形成增加。
2.Pharmacodynamics
The pharmacodynamics of mitotane are unknown.
3.Pharmacokinetics
Absorption
After oral administrationmitotane40%of the dose is absorbed.
Assignment
Mitotan is found in most tissues of the body; however, fat is the predominant site of distribution.
Elimination
米托坦停药后,血浆终末半衰期为18至159天(中值为53天)。
Metabolism
Mitotan is converted to water-soluble metabolites.
Excretion
Unaltered mitotane was not found in urine or bile. Approximately10% of the administered dose is recovered in the urine as water-soluble metabolites. Varying amounts of metabolites(1%-17%)are excreted into bile.
There are generic drugs that are officially on the market in Laos. The retail price is about 500mg100 tablets3 about one thousand. If you want to get more high-quality information, you can contact Yaode. Yade will do its best to learn more about high-quality overseas drugs for you.
xa0
xa0
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















