What medicine is Leqselvi (Deuruxolitinib)?
Leqselvi (Deuruxolitinib) is a Janus kinase (JAK) inhibitor indicated for the treatment of severe alopecia areata in adults. Alopecia areata is an autoimmune disease in which the immune system attacks hair follicles, causing partial or total hair loss on the scalp and body. Leqselvi contains deuruxolitinib, a JAK1/JAK2 inhibitor that reverses hair loss by targeting the γc cytokine and interferon-γ (IFN-γ) signaling pathways to treat alopecia areata.
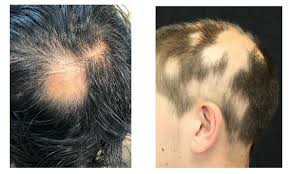
The FDA's approval of Leqselvi was based on data from the Phase 3 clinical trials THRIVE-AA1 and THRIVE-AA2, which evaluated scalp hair regrowth after 24 weeks, and two long-term extension trials in which patients were eligible after completing 24 weeks of the trial. At study baseline, the average patient had only 13% scalp hair coverage. In the trial, the primary endpoint was that more than 30% of patients taking Leqselvi achieved scalp hair coverage of 80% or greater at 24 weeks. Additionally, up to 25% of patients had ≥90% scalp hair coverage at 24 weeks.
Leqselvi tablets are taken by mouth twice daily. Common adverse reactions include headache, acne, nasopharyngitis, elevated blood creatine phosphokinase, hyperlipidemia, fatigue, weight gain, lymphopenia, thrombocytosis, anemia, skin and soft tissue infections, neutropenia, and herpes. The Leqselvi product label has a warning box warning of serious infections, higher all-cause mortality, higher risk of malignancies (such as lymphoma and lung cancer), higher rates of major adverse cardiovascular events (MACE), and higher rates of thrombosis. Warnings and precautions include gastrointestinal perforation and certain laboratory abnormalities.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















