Is there a 500mg strength for Margituximab/Meganex?
Margetuximab is a new generation of anti-HER2-targeting monoclonal antibody, which is used in the treatment of malignant tumors such as HER2-positive advanced breast cancer. When patients and family members are concerned about the use of drugs, they often ask whether the specifications include the 500mg version, so as to understand the amount of medication and financial expenditure. According to the current public information, the original drug of margituximab has been launched in China. However, due to the short time to market, the specifications and price system are still being gradually improved. At present, the common domestic market specification is 250mg/10ml, 1 bottle per box. Overseas, Europe and Japan have approved the drug, and the supplied specifications are also mainly 250mg. There is no independent specification of 500mg.
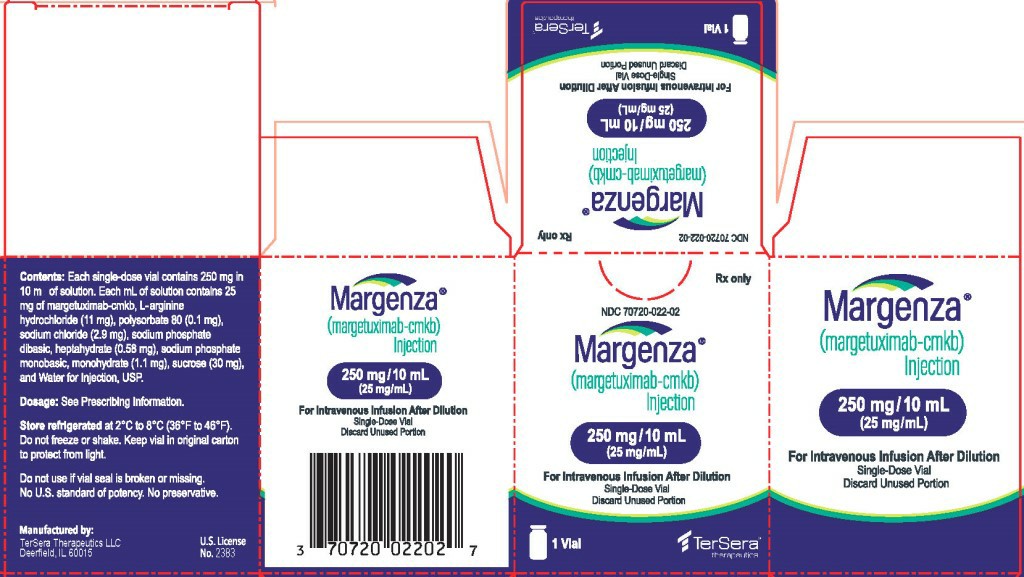
Drug dosage design and specification setting usually consider the flexibility and safety of clinical use. For monoclonal antibody drugs, the packaging specification of 250mg/bottle can better meet the individualized dosing needs of patients of different weights. Since the recommended dose of margetuximab is calculated based on body weight (15 mg/kg), using 250 mg as a distribution unit can meet the different dosage needs of patients by combining different numbers of vials in actual clinical practice. This design method not only facilitates preparation, but also avoids drug waste. If there is a large format packaging of 500 mg, although it is possible to reduce the number of bottles used by some overweight patients, it is not conducive to precise dosage adjustment in most patient groups.
In terms of storage and transportation, magituximab is a protein preparation and is sensitive to both temperature and light. Drugs in small packages can reduce the risk of loss during distribution, transportation and storage, and are more in line with the medication management habits of hospital pharmacies and clinical departments. Therefore, both the Chinese market and overseas markets currently use 250mg as the main specification, and no independent packaging of 500mg has been launched.
Reference materials:https://www.margenza.com/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















