Interpretation of the efficacy and price of Sotorasib: a new breakthrough in KRAS targeting that breaks the "undruggable"
1. From“impossible” to “possible”: KRASThe historical turning point of the target
In the history of cancer treatment, KRAS mutations have always been regarded as "undruggable targets." In the past 40 years, countless pharmaceutical companies have invested huge amounts of money, but have repeatedly failed. KRASThe protein structure encoded by the KRAS gene is tight and smooth, with almost no obvious "binding pockets". This made scientific researchers once think that it is like a "polished marble" and cannot be locked by drugs at all.
However, scientific breakthroughs often occur in desperate times. In 2013, researchers discovered the special weakness of the KRAS G12C mutation and successfully designed a small molecule inhibitor that can "block" the mutant protein. Sotorasib (Sotorasib, trade name: Lumakras) was born and received accelerated approval from the USFDA in 2021 for the treatment of KRAS Patients with G12Cmutated non-small cell lung cancer (NSCLC).
This incident not only changedKRAS’s historical evaluation of “undruggable”, but also opened up a new field for precision medicine.

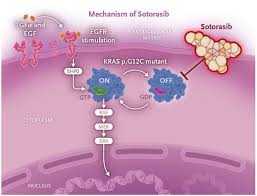
2. The mechanism of action of sotorasibu: precision“Locked”KRAS G12C
Sotoraxib is an oral small molecule inhibitor that can specifically bind to the KRAS proteinG12C mutation site, locking KRAS in an inactive state and blocking cancer cell proliferation signals.
Different from traditional chemotherapy"indiscriminate bombing", the target of sotoracib is very precise. It is like a key and can only open "G12C"Mutation" is a specific lock. Therefore, only patients who are confirmed by molecular testing to carry the KRAS G12C mutation may benefit from it.
Globally, KRAS G12C mutations account for approximately NSCLC 13%-14% of patients, which means that the potential applicable population of sotoraxib is not small.

3. Clinical efficacy: Enlightenment from real-world data
The efficacy of sotoraxib mainly comes from CodeBreaK 100 clinical trials. This is a multicenter phase II study evaluating sotorasib in KRAS G12C mutated NSCLC.
1. Effectiveness Performance
Objective response rate (ORR): 37%
Disease control rate (DCR): 80%
Median progression-free survival (PFS): 6.8months
Median overall survival (OS): 12.5months
Such results are quite rare among patients who have failed multiple lines of treatment in the past.
2. Security
Common adverse reactions: diarrhea, nausea, elevated transaminase, joint pain.
Most of them are level 1-2 and are highly controllable.
3. Follow-up research
CodeBreaK 200Study shows: In second-line treatment, sotoraxib significantly improves PFSPFS compared with docetaxel, and the side effects are more controllable.
Combination regimens with immunotherapy or other targeted drugs are becoming a new direction.
These data suggest that sotorasiib brings new therapeutic hope to patients with KRAS G12C mutations.
4. Price and market: expensive original drugs and affordable generic drugs
Although the efficacy is good, drug price is always the most sensitive issue for patients.
1. Domestic Current Situation
As of2025, sotoraxibu has not yet been officially launched in mainland China.
Patients cannot purchase drugs directly through regular domestic hospitals and can only choose to purchase drugs overseas or go abroad.
2. Overseas price situation
European version and Hong Kong version of original medicine: a box of medicine often costs tens of thousands of yuan. Long-term medication costs may be as high as hundreds of thousands of yuan.
The annual treatment cost in the U.S. market is as high as hundreds of thousands of dollars, which is almost unaffordable for ordinary patients.
3. The emergence of generic drugs
Two companies in Laos - Lucius and Daxiong, have successfully produced generic versions of sotorasibu.
The latest price:120mg*56The specifications of the tablets only cost more than one thousand yuan, dozens of times cheaper than the original drug.
The ingredients are basically the same as the original drugs, which undoubtedly relieves patients from huge financial pressure.
4. The meaning behind the price
The high pricing of original drugs reflects the huge cost of research and development of innovative drugs; while the low prices of generic drugs reflect the social value of "drug accessibility". For patients, reasonable and legal access to affordable drugs is the real key to saving lives.

5. Three key issues in patients’ medication use
1. Am I suitable to use Sotoraxibu?
Only applicable to KRAS G12C mutation-positive patients.
The target must be confirmed through molecular testing such as NGS or PCR.
2. How to weigh original drugs and generic drugs?
Original drug: sufficient data and high degree of protection, but the cost is extremely high.
Generic drugs: They are cheaper and have the same ingredients, but you need to ensure that they come from a legitimate source to avoid buying fake drugs.
3. Drug risk and management
Regularly review liver function and monitor adverse reactions.
Strictly follow the doctor's instructions and do not stop or increase the dosage on your own.

6. Future Outlook:KRASThe "first step" of inhibitors
The advent of sotorasiib marks the "first step" in KRAS targeted therapy, but it is by no means the end.
MoreKRASInhibitors are about to appear: new drugs such asAdagrasib have been approved in Europe and the United States and may enter the Chinese market.
Combination therapy research: Combination with EGFR inhibitors and immunotherapy may further improve the efficacy.
Medical insurance potential: Once sotorasibu is launched in China and entered into medical insurance, the financial burden on patients will be significantly reduced.
Real-world data accumulation: As the scope of application expands, more clinical practice data will support its use value in the future.
Sotoracib is a landmark drug that breaks the myth that KRAS is "undruggable" and gives lung cancer patients with G12C mutations hope for precise treatment for the first time.
However, the real problems are equally acute: original drugs are expensive and have not yet been launched in the country; generic drugs are cheap but have complicated channels. For patients, scientific testing, rational choices, and regular drug purchase are the guarantee of life.
In the future, as moreKRAS inhibitors come on the market, medical insurance policies are gradually improved, and the generic drug market is standardized, sotorasibu will surely benefit more patients. It is not only a medicine, but also a signal: Precision medicine is constantly breaking through the "impossible" in the past.
xa0
References
1.Skoulidis F, et al. Sotorasib for Lung Cancers with KRAS p.G12C Mutation. N Engl J Med. 2021.
2.Amgen. Lumakras (sotorasib) prescribing information. https://www.lumakras.com
3.ClinicalTrials.gov. CodeBreaK 100 and CodeBreaK 200 Studies. https://clinicaltrials.gov
4.Center for Drug Evaluation of the State Food and Drug Administration. http://www.cde.org.cn
5.Interpretation of the National Medical Insurance Administration Drug Catalog. http://www.nhsa.gov.cn
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















