What are the main manufacturers of selumetinib?
Selumetinib (Selumetinib) is a MEK inhibitor, and its research and development background, supply channels and manufacturers have always been the focus of patients' attention. As it is widely recognized internationally and gradually enters the marketing system of many countries, the production pattern of its original drugs and generic drugs has become increasingly clear. In domestic and foreign markets, the original drug selumetinib has been approved for sale in many regions, and generic drugs with stable quality and consistent ingredients have gradually emerged, providing patients with more treatment options.
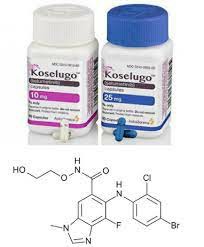
The original drug selumetinib was developed by a large international pharmaceutical company. It was the first version to enter the global market and complete a number of key studies. Its products have strict standards in terms of technology, stability, and purity, so they are used in clinical applications as the first-choice marketed drugs in many countries. It is worth noting that the original drug selumetinib has been officially launched in China, allowing domestic patients to obtain the drug without relying on cross-border channels, while also ensuring quality traceability and drug safety.
In overseas markets, the European version of selumetinib’s original drug has also been on the market for many years and has become a routinely supplied treatment option in the medical systems of many countries. Due to differences in regulatory systems in different regions, drug approval standards may be slightly different, but the ingredients, specifications and safety of the original drugs remain consistent to ensure the reliability of clinical use.
As the application scope of selumetinib expands, pharmaceutical companies in many countries have begun to produce its generic drugs. Currently known sources of generic drugs include Lao Lucius Pharmaceuticals, Lao Daxiong Pharmaceuticals and other companies. The generic versions of selumetinib produced by them are basically the same as the original drugs in terms of ingredients, dosage forms and administration methods, aiming to provide patients who require long-term medication with a more cost-effective option. Many generic drug companies have adopted international production standards to ensure that the bioequivalence of drugs meets regulatory requirements, and have gradually entered legal channels in some countries.
Reference materials:https://www.koselugo.com
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















