Resistance management of Sarilumab
Rheumatoid arthritis(RA) is a chronic systemic inflammatory disease that mainly affects peripheral joints. Sarilumab is a human monoclonal antibody against IL-6 receptor-α. In Phase II and III clinical trials, salirumab in the context of methotrexate demonstrated superior clinical efficacy to placebo in patients with rheumatoid arthritis who had an inadequate response to methotrexate or an inadequate response to or intolerance to TNF inhibitors.
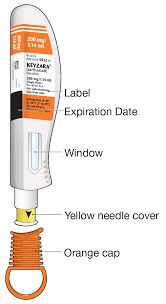
Salirumab monotherapy has also been shown to be superior to adalimumab monotherapy in patients with rheumatoid arthritis who have an inadequate response to or are intolerant to methotrexate. In terms of safety, injection site reactions, neutropenia, and increases in liver enzymes and serum cholesterol were more common in the salirumab group than in the placebo group. Overall, salirumab has the potential to be another effective antirheumatic drug against active rheumatoid arthritis. Salirumab may help control the symptoms of the condition, but it will not cure it. Continue taking salivolumab even if you feel well. Do not stop using Salirumab without consulting your doctor. Everyone has a different body constitution and different drug resistance reactions. If you feel unwell, please inform your doctor in time.
The patent drug of Salirutumab is a new type of drug for treating diseases. It is not yet on the market in China, and therefore it is not covered by medical insurance. Salirutumab The common dosage form of the original drug is injection, which has been marketed overseas. The European version of the specification200mg*2 per box is priced around 17,500 (The price may fluctuate due to the exchange rate). The purchase of the original drug of Salirutumab needs to be purchased through formal channels. There are currently no generic versions of Salirutumab produced in other countries.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















