Efficacy and effects of Sarilumab
Three studies involving more than 2,100 adults with rheumatoid arthritis show that after 24 weeks of treatment, Sarilumab can effectively reduce joint pain and swelling, improve joint motion, and slow joint damage. The first study involved approximately 1,200 patients who had an inadequate response to methotrexate treatment and received either salivolumab plus methotrexate or placebo plus methotrexate. Based on the standardized score (ACR20), 58% of patients given 150 mg of salirumab and 66% of patients given 200 mg of salirumab experienced a 20% or greater reduction in symptoms.
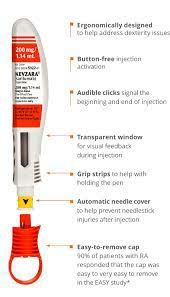
The second study involved546 patients whose condition did not respond adequately to or could not take TNF-alpha inhibitors (another drug used to treat rheumatoid arthritis); all patients received salirumab or a placebo combined with DMARDs. Fifty-six percent of patients who received salivolumab 150 mg and 61% of patients who received 200 mg experienced a 20% or greater reduction in symptoms. The third study, involving 369 patients, compared salirumab with adalimumab (another monoclonal antibody used to treat rheumatoid arthritis). Patients who received salirumab had greater improvements in joint function (based on a standardized score called the DAS28-ESR) than those who received adalimumab.
The patent drug Thalilutumab is a new type of drug for treating diseases. It is not yet on the market in China, and therefore it is not covered by medical insurance. The common dosage form of Salirutumab's original drug is injection, which has been marketed overseas. The price of the European version of the specification200mg*2 per box is around 17,500 (the price may fluctuate due to the exchange rate). There are currently no generic Salirutumab produced in other countries.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















