Rezlidhia (Olutasidenib) Efficacy
The U.S. Food and Drug Administration (FDA) approved Rezlidhia (olutasidenib) capsules for use in adult patients with relapsed or refractory acute myeloid leukemia who have a predisposing IDH1 mutation detected by an FDA-approved test. Approval was based on Study 2102-HEM-101, an open-label, single-arm, multicenter clinical trial that included 147 adult patients with relapsed or refractory acute myeloid leukemia and confirmed IDH1 mutations using the above-described assay.
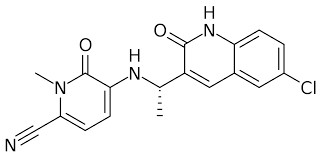
In clinical trials, patients received Rezlidhia 150 mg orally twice daily until disease progression, unacceptable toxicity, or hematopoietic stem cell transplantation. The median duration of treatment was 4.7 months, and 16 patients received Rezlidhia post-hematopoietic stem cell transplantation. Efficacy was determined based on the ratio of complete remission plus complete remission with partial hematologic recovery, the duration of complete remission plus complete remission with partial hematologic recovery, and the rate of conversion from transfusion dependence to independence. The rate of complete remission plus complete remission with partial hematological recovery was 35%, including 32% complete remission and 2.7% complete remission with partial hematological recovery. The median duration of complete remission plus complete remission with partial hematologic recovery was 1.9 months, and the median duration of complete remission plus complete remission with partial hematologic recovery was 25.9 months.
Of the 86 patients who were red blood cell and/or platelet transfusion dependent at baseline, 29 became red blood cell and platelet transfusion independent during any 56-day period after baseline. Of the 61 patients who were red blood cell and platelet transfusion independent at baseline, 39 remained transfusion independent 56 days after baseline.
After Rezlidhia is approved for marketing, there is little information about its price and other related information. Please consult the medical consultant for details.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















