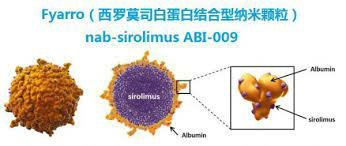
How to administer sirolimus albumin-bound nanoparticle injection (FYARRO)
The recommended dose of sirolimus albumin-bound nanoparticle injection (FYARRO) is 100 mg/m2 as a 30-minute intravenous infusion on days 1 and 8 of each 21-day cycle until disease progression or unacceptable toxicity. If a patient experiences adverse reactions after infusion of sirolimus albumin-bound nanoparticle injection, the first dose should be reduced to 75 mg/m2 (25% of the recommended dose), the second dose should be reduced to 56 mg/m2 (25% of the first reduced dose), and the third dose should be reduced to 45 mg/m2 (25% of the second reduced dose).
Patients who are intolerant after three dose reductions should permanently discontinue sirolimus albumin-bound nanoparticle injection. Patients with mild hepatic impairment (total bilirubin ≤ ULN, AST > ULN, or total bilirubin > 1-1.5 × ULN, any AST) need to reduce the dose of sirolimus albumin-bound nanoparticle injection to 56 mg/m2. Patients with moderate hepatic impairment (total bilirubin >1.5-3.0 × ULN, any AST) require a dose reduction to 75 mg/m2. Avoid use in patients with severely impaired hepatic function.
The original drug of sirolimus albumin-bound nanoparticle injection has not yet been marketed in China, so it is not included in medical insurance. The US version of sirolimus albumin-bound nanoparticle injection Original drug, specifications The price of each 100mg tube may be around RMB 60,000 (the price may fluctuate due to the exchange rate), which is relatively expensive. Currently, there is no generic version of Sirolimus Albumin-Bound Nanoparticle Injection on the market. For more drug information and specific prices, please consult Yaode Medical Consultants.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















