Is Toripalimab on the market?
Due to the satisfactory anti-tumor effect of Toripalimab (Toripalimab) in nasopharyngeal carcinoma (NPC) , the U.S. Food and Drug Administration (FDA) also granted it orphan designation on May 18, 2020 and sold it under the trade name LOQTORZI. Due to the superior clinical benefits conferred by the combination of toripalimab and gemcitabine-cisplatin (GP), the National Medical Products Administration has recently approved toripalimab plus GP as a first-line treatment for nasopharyngeal carcinoma, marketed under the trade name Tuoyi.
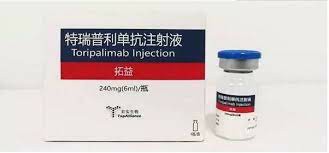
Toripalimab Injection is a clear to slightly opalescent, colorless to slightly yellow solution packaged in a carton containing a 240 mg/6 ml (40 mg/ml) single-dose vial. Toripalimab is the first anti-PD-1 monoclonal antibody approved by the National Medical Products Administration (10). It consists of two 452-amino-acid heavy chains and two 219-amino-acid light chains, and contains an N-linked glycosylation site at N302 of each heavy chain. Toripalimabcan bindPD-1 and effectively block the interaction with its ligand; the blocking is mainly attributed to the stereospecific hindrance of the heavy chain.
Immune-mediated adverse reactions have been reported with toripalimab, including pneumonitis, colitis, hepatitis, endocrine disorders, renal insufficiency nephritis, and cutaneous adverse reactions. The most common adverse reactions of toripalimab in combination with cisplatin and gemcitabine are nausea, vomiting, decreased appetite, constipation, hypothyroidism, rash, pyrexia, diarrhea, peripheral neuropathy, cough, musculoskeletal pain, upper respiratory tract infection, insomnia, dizziness, and malaise. The most common adverse effects of toripalimab monotherapy were fatigue, hypothyroidism, and musculoskeletal pain.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















