Avonib Instructions for Avonib
一.Common name:ivosidenib
Product name:Tibsovo
All names:ivosidenib,ivosidenib,ivosidenib,Tibsovo
2. Indications:
1.Newly diagnosed acute myeloid leukemia
Ivonib is indicated for the treatment of newly diagnosed acute myeloid leukemia (AML) in adult patients ≥75 years of age or with comorbidities that preclude the use of intensive induction chemotherapy, as tested in an FDA-approved trial.
2.Relapsed or refractory acute myeloid leukemia
Ivonib is indicated for the treatment of adult patients with relapsed or refractory acute myeloid leukemia(AML)who have a susceptibility to isocitrate dehydrogenase-1 (IDH1)mutated by aFDAapproved test.
Three. Dosage and usage
1.Recommended dosage
The recommended dose of ivonib is 500 mg orally once daily until disease progression or unacceptable toxicity. For patients without disease progression or unacceptable toxicity, treat for at least 6 months to allow time for clinical response.
Missed dose:
If a dose of ivonib is missed or not taken at the usual time, the dose should be taken as soon as possible and at least 12 hours before the next scheduled dose and return to your normal schedule the next dayDo not take 2 doses within 12 hours.
2.Dose adjustment when using strongCYP3A4 inhibitors
If coadministration of strong CYP3A4inhibitors is necessary, reduce ivonib dose to 250 mg once daily. If a strong inhibitor is discontinued, increase the ivonib dose(after at least 5 half-lives of the strong CYP3A4 inhibitor) to the recommended dose 500 mg, once daily.
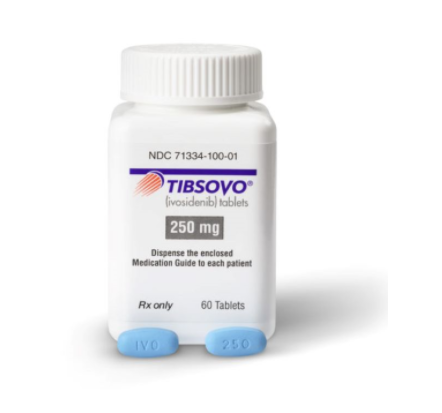
4. Dosage Form and Strength
Tablets: 250mg, blue oval film-coated tablets, with "IVO" debossed on one side and "250" debossed on the other side.
Five. Contraindications
None
6. Warnings and Precautions
1.Differentiation Syndrome
Patients who receivedivonib developed symptoms of differentiation syndrome, which can be fatal if left untreated. Symptoms may include fever, dyspnea, hypoxia, pulmonary infiltrates, pleural or pericardial effusion, rapid weight gain or peripheral edema, hypotension, and hepatic, renal, or multiorgan dysfunction. If differentiation syndrome is suspected, initiate corticosteroid therapy and hemodynamic monitoring until symptoms resolve.
2.QTcInterval prolongation
Patients receiving ivosidenib may experience QT (QTc) prolongation. Avosidenib is combined with drugs known to prolong the QTc interval(such as antiarrhythmic drugs, fluoroquinolones, triazole antifungals, 5-HT3receptor antagonists) and CYP3A4 inhibitors may increase QTcRisk of interval prolongation, monitor ECG and electrolytes. More frequent monitoring may be needed in patients with congenital long QTc syndrome, congestive heart failure, electrolyte abnormalities, or who are taking drugs known to prolong the QTc interval. If QTcincreases to greater than 480ms to less than 500ms, discontinue ivonib. If QTcincreases beyond 500 milliseconds, interrupt and reduce ivonib. Ivonib should be permanently discontinued in patients who develop QTc prolongation with signs or symptoms of life-threatening arrhythmias.
3.Guerin-Barré Syndrome
In this clinical study, the incidence of Guillain-Barré syndrome was less than1% (2/258) among patients treated with ivonib. Monitor patients takingivosidenib for new signs or symptoms of motor or sensory neuropathy, such as unilateral or bilateral weakness, sensory changes, paresthesia, or dyspnea. Patients diagnosed with Guillain-Barré syndrome should permanently discontinue ivonib.
7. side effects
The most common adverse reactions(≥20%)are fatigue, arthralgia, leukocytosis, diarrhea, edema, nausea, dyspnea, mucositis, electrocardiogramQTprolongation, rash, cough, decreased appetite, myalgia, constipation and fever.
The most common laboratory abnormalities (≥20%)are decreased hemoglobin, decreased calcium, decreased sodium, decreased magnesium, increased uric acid, decreased potassium, increased alkaline phosphatase, increased aspartate aminotransferase, decreased phosphate, increased creatinine.
Clinical Pharmacology
1.Mechanism of action
Ivonib is a small molecule inhibitor of mutant isocitrate dehydrogenase 1 (IDH1) enzyme. Ivosidenibinhibits selected IDH1 R132mutants in vitro at much lower concentrations than wild-type IDH1. InIDH1 mutatedAMLIn a mouse xenograft model, ivonibinhibition of mutated IDH1enzyme resulted in reduced 2-HG levels and induction of myeloid differentiation in vitro and in vivo. In blood samples from IDH1mutated AML patients, ivonib reduced 2-HG levels in vitro, reduced blast counts, and increased the percentage of mature myeloid cells.
2.Pharmacodynamics
Multiple daily doses of 500 mgof ivonib were observed to reduce plasma 2-HG concentrations in patients with hematological malignancies to levels similar to baseline levels in healthy subjects and reduced 2-mercury concentrations in the bone marrow by more than 90%.
3.Pharmacokinetics
Absorption
The median maximum time to reach C is approximately 3 hours.
The influence of food
After a single dose in healthy subjects, a high-fat meal(approximately900 to 1000 calories, 500 to 600 span>Fat calories, 250Carbohydrate calories and 150Protein calories )IncreasedAvonib CMaximum98% (90% confidence interval: 79%, 119%) and AUC for intermediate-range nuclear forces are approximately 25%.
DistributionDistribution
The mean apparent volume of distribution at steady state is 234 L (%CV: 47). The in vitro protein binding rate of ivonib is 92-96%.
Elimination
The terminal half-life of ivonib is 93 hours (%CV: 67), the apparent clearance rate(CL/F) is 4.3L/hours(%CV: 50).
Metabolism
Ivonibis the major component (> 92%) of the total radioactivity in plasma. Avosidenib b is mainly metabolized by CYP3A4 and to a lesser extent by N- dealkylation and hydrolysis pathways.
Excretion
After a single oral administration of radiolabeledivosidenibto healthy subjects,77%ofivosidenib was eliminated in feces (67%unchanged), 17% eliminated in urine (10%no change).
9. Used by specific groups of people
1.Pregnancy
Based on animal embryo-fetal toxicity studies, pregnant women taking TIBSOVO may cause harm to the fetus. There are no available data regarding use of TIBSOVO in pregnant women to inform about the drug-related risks of major birth defects and miscarriage. In animal embryonic fetotoxicity studies, oral administration of ivonib to pregnant rats and rabbits during organogenesis was associated with fetal toxicity based on AUC at recommended human doses. span>Fetal mortality is associated with changes in growth starting at 2 times the steady-state clinical exposure.If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, inform patients of the potential risk to the fetus.
2.Breastfeeding
There are no data on the presence of ivonib or its metabolites in breast milk, the effects on breastfed infants, or the effects on milk production. Because many drugs are excreted in breast milk and breastfed children may experience adverse reactions, it is recommended that women receiveivonibDo not breastfeed during treatment and for at least 1month after the last dose.
3.Pediatric use
The safety and effectiveness of TIBSOVO in pediatric patients have not been established.
4.Medication for the Elderly
In clinical studies, 34of AML patients were newly diagnosed with 33 (97%) ages 65years old or above, 19patients(56%)age 75years or above. In clinical studies, 112 of 179 (63%) patients with relapsed or refractory AML (63%) had relapsed or refractory AML. an>age 65 years or above, 40cases(22%)age 75 years or above. No overall differences in efficacy or safety were observed between patients ≥65 years of age with relapsed or refractory AML and older and younger patients.
5.Renal function impairment
For mild or moderate renal impairment (eGFR ≥ For patients with 30ml/minutes/1.73m), it is not recommended to modify the starting dose2, MDRD). The pharmacokinetics and safety of ivonib 2MDRD) are unknown in patients with severe renal impairment (eGFR < 30 mL/min/1.73m) or requiring dialysis. In patients with severe renal impairment or who require dialysis, consider the risks and potential benefits before starting treatment with TIBSOVO.
6.Liver function impairment
For mild or moderate(Child-Pugh A grade or B grade)It is not recommended to modify the starting dose in patients with liver function impairment. The pharmacokinetics and safety of ivonib in patients with severe hepatic impairment (Child-Pugh C grade) are unknown. In patients with pre-existing severe hepatic impairment, consider the risks and potential benefits before initiating treatment with TIBSOVO.
Avonibu produced in Laos costs about 5,000 to 6,000 yuan per box, and the price is favorable. If you want to get more high-quality information, you can contact Yaode . Yaode will do its best to learn more about high-quality overseas drugs for you.
xa0
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















