Instructions for enasidenib
1. All names: Enasidenib,Idhifa,Enasidenib
Common name: Ensidipine
Product name:Idhifa
2. Indications and Usage
Ensidipine is an isocitrate dehydrogenase2 inhibitor indicated for the treatment of isocitrate dehydrogenase2 detected in an FDA-approved test. (IDH2)mutated relapsed or refractory acute myeloid leukemia(AML)Adult patients.
Three.Dosage and usage
100 mg orally once daily until disease progression or unacceptable toxicity occurs.For patients without disease progression or unacceptable toxicity, treat for at least 6 months to allow time for clinical response.
4. Dosage Form and Strength
Tablets: 50mg or 100mg
Five. Contraindications
None
6. Warnings and Precautions
1.In clinical trials, 14%of patients receiving ensidipine developed differentiation syndrome, which may be life-threatening or fatal if left untreated. Differentiation syndrome is associated with rapid proliferation and differentiation of myeloid cells. Although there are no diagnostic tests to identify the syndrome, symptoms in patients treated with ensidipine included acute respiratory distress manifested by dyspnea or hypoxia (68%) and the need for supplemental oxygen (76%) Lymphadenopathy (33%); bone pain (27%); peripheral edema with rapid weight gain (21%); and pericardial effusion (18%). Hepatic, renal, and multi-organ dysfunction was also observed. A differentiation syndrome with or without hyperleukocytosis was observed between 1 days and 5 months after initiation of ensidipine. If differentiation syndrome is suspected, initiate oral or intravenous corticosteroids (eg, 10 mg of dexamethasone every 12 hours) and monitor hemodynamics until symptoms improve. Taper corticosteroids only after symptoms resolve. Premature discontinuation of corticosteroid therapy may result in recurrence of differentiation syndrome symptoms. If severe pulmonary symptoms or renal insufficiency requiring intubation or ventilator support persist more than 48 hours after initiation of corticosteroids, discontinue ensidipine until signs and symptoms are no longer severe. Hospitalization is recommended for close observation and monitoring of patients with pulmonary,/or renal manifestations.
2.Embryo-Fetal toxicity. Based on animal embryo-fetotoxicity studies, ensidipine can cause embryo-fetal damage when administered to pregnant women. In animal embryo-fetotoxicity studies, the area under the concentration-time curve(AUC) at the recommended human dose ), Embryo-fetotoxicity due to ensidipine begins at 0.1 times the steady-state clinical exposure. Inform pregnant women of potential risks to the fetus. Advise females of reproductive potential to use effective contraception during treatment with ensidipine and for at least 2 months after the last dose. Advise men who are partners of a female of reproductive potential to use effective contraception during treatment with ensidipine and for at least 2 months after the last dose.
7. adverse reactions
The most common adverse reactions (≥20%) are nausea, vomiting, diarrhea, increased bilirubin and decreased appetite.
Eight.Drug Interactions
1.EnsidipineEffects on other drugs
OATP1B1, OATP1B3 and BCRPsubstrates
Ensidipine is an OATP1B1, OATP1B3, and BCRP inhibitor. Coadministration with ensidipine increases exposure to OATP1B1, OATP1B3 and BCRP substrates, which may increase the incidence and severity of adverse reactions with these substrates. Reduce the dose of OATP1B1, OATP1B3 and BCRP substrates as recommended and clinically indicated in the corresponding prescribing information.
2.SomeP-glycoprotein substrates
Ensidipineis aP-gpinhibitor. Coadministration with ensidipine increases the exposure of P-gp substrates, which may increase the incidence and severity of adverse reactions with these substrates .For small concentrations Changes in P-gpsubstrates that may result in serious adverse reactions, reduce the dose or modify the dosing frequency of such P-gpsubstrates and monitor for adverse reactions as recommended in the appropriate prescribing information.
9. Used by specific groups of people
1.Pregnancy. May cause harm to unborn baby. Confirm pregnancy status before initiating treatment with ensidipine. If used during pregnancy, or if the patient or their partner becomes pregnant during treatment, inform patients of the potential fetal risk.
2.Breastfeeding. It is unknown whether ensidipine is distributed in breast milk. Stop nursing during treatment and ≥2 months after discontinuation of treatment.
3.Females and men of reproductive potential. Females of reproductive potential and men who are partners of such women during and after drug treatment≥2Appropriate contraceptive methods should be used within months. Animal studies suggest that ensidipine may impair fertility in both women and men.
4.There is generally no difference in safety and effectiveness when used by the elderly compared with young people.
5.Liver damage. In a population pharmacokinetic analysis, mild hepatic impairment did not alter systemic exposure. However, exposure may be increased because ensidipine is primarily metabolized by the liver.
6.Kidney damage. In population pharmacokinetic analyses, renal impairment did not alter systemic exposure.
10. Description
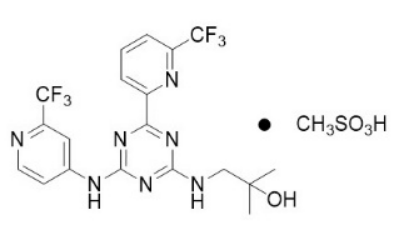
Ensidipineis an inhibitor of isocitrate dehydrogenase-2 (IDH2)enzyme. Enalapril is available as the mesylate salt with the chemical name: 2-methyl-1-[(4-[6-(trifluoromethyl)pyridine< /span>-2-yl]-6-{[2-(trifluoromethyl)pyridine-4-base]amino}-1, 3, 5-triazine -2-yl)amino]propanol-2-alcohol methanesulfonate.
The chemical structure is:
11. clinical pharmacology
1.Mechanism of action
Ensidipine is a small molecule inhibitor of isocitrate dehydrogenase 2 (IDH2) enzyme. In vitro, ensidipinetargets mutantIDH2 at a concentration approximately40fold lower than the wild-type enzyme.VariantsR140Q, R172S and R172K. In a IDH2mutatedAML mouse xenograft model,ensidipine suppresses the mutantIDH2enzyme causes a decrease in 2-hydroxyglutarate(2-HG) levels and induces myeloid differentiation in vitro and in vivo. In blood samples from IDH2mutated AML patients, ensidipine reduced 2-HG levels, reduced blast counts, and increased the percentage of mature myeloid cells.
2.Pharmacokinetics
Peak plasma concentration(CMaximum) after a single dose of 100mg is 1.4microgram/ml[Coefficient of variation(CV % 50%], under steady state daily 100mg is 13.1micrograms/ml(CV% 45%)The area under the concentration-time curve of ensidipine (AUC) ranges from 50 in an approximate dose proportional manner. mg (0.5 times ) increased to 450 mg (4.5 times) after once daily dosing. Steady-state plasma levels are reached within 29 days. When administered once daily, accumulation is approximately 10 times.
3.Absorption
The absolute bioavailability of ensidipine is approximately57% after oral administration of a 100 mg dose. After a single oral dose, C is achievedThe median time to max(Tmax) is 4 hours.
4.Allocation
The average volume of distribution of ensidipine(Vd) is 55.8liters(CV% 29). In vitro, the human plasma protein binding rate of ensidipine is 98.5%, and the binding rate of its metabolite AGI-16903 is 96.6%.
5.Elimination
The terminal half-life of ensidipine is 7.9 days, and the average systemic clearance rate (CL/F) is 0.70liters/ hours(CV% 62.5).
6.Metabolism
Ensidipine accounts for 89% of circulating radioactivity, and AGI-16903, the N-dealkylation metabolite, accounts for 10% of circulating radioactivity.
In vitro metabolism of ensidipine by multiple cytochromesP450 (CYP)Enzymes(For example, CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6
7.Excretion
Eighty-nine percent(89%)of ensidipine is eliminated in feces and 11% is eliminated in urine. Excretion of unchanged ensidipine in feces represents 34% of the radiolabeled drug and 0.4% in urine.
8.Specific groups of people
No clinically meaningful effects on ensidipine pharmacokinetics were observed among the following covariates:Age(19years to100years), race(White, black, or Asian), mild liver function impairment[defined as total bilirubin ≤ upper limit of normal(ULN)and aspartate aminotransferase(AST) >ULNor total bilirubin1 to 1.5 timesULN and anyAST], renal impairment(Cockcroft-Gault’s formula is defined as creatinine clearance ≥30 mL/min), gender, body weight (39 kg to 136 kg), and body surface area.
9.Drug interaction studies
clinical research
OATP1B1, OATP1B3 and BCRPSubstrates:Multiple DoseIDHIFA 100 mg followed by rosuvastatin10 mgcan increase rosuvastatinCmaximum366% and AUC0-INF increased by 244%.
P-gpSubstrate:Coadministration of digoxin0.25 after multiple dosesIDHIFA 100 mg mgincreased digoxinCmaximum26% and AUC0-30hours decreased 20%.
The retail price of Ziska Pharmaceuticals in Bangladesh50mg60tablets4Around one thousand yuan, the retail price of Everest Pharmaceuticals in Bangladesh50mg30 Pellets5Around 1,000 yuan, the retail price of Lucius Pharmaceuticals in Laos is 2about 1,000 yuan, and all have government approvals. Patients can choose the drug version according to their own financial situation. If you want to get more high-quality information, you can contact YaDE. YaDE will do its best to learn more about high-quality overseas drugs for you.
xa0
xa0
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















