What is the price of pemetinib/pemetinib after it is launched in China?
In mainland China,pemetinib/pemetinib(Pemigatinib) is approved for the systemic treatment of unresectable locally advanced or metastatic cholangiocarcinoma limited to chemotherapy. The standard of first-line treatment for locally advanced or metastatic cholangiocarcinoma is gemcitabine plus cisplatin; however, this provides limited benefit as most patients will experience disease progression within 1 year of treatment. Pemetinib is an oral, potent and selective inhibitor of FGFR1, FGFR2 and FGFR3. It is the first targeted therapy drug for cholangiocarcinoma approved by the United States, the European Union, Japan and Taiwan, and is suitable for cholangiocarcinoma that has previously received FGFR2 fusion or rearrangement therapy.
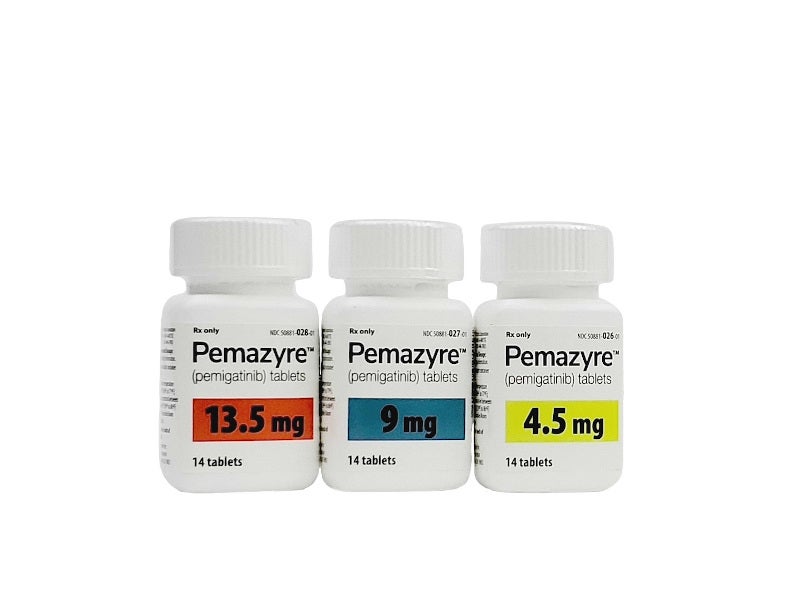
Pemetinib is an innovative drug targetingFGFR variant cholangiocarcinoma. Its unique treatment mechanism and significant clinical effects provide new treatment hope for these patients. However, its use still needs to be done with caution and under the guidance of a professional doctor. The original drug has been Pematinib tablets(Dabotan/PEMAZYRE) are now available in ChinaListed in China,Butdue to the short listing time,stillcannotto enter the medical insuranceRank, The ones sold in China are4.5mg*14 tablets, 9mg*14 tabletsTwo kindsSpecifications The price of each box of medicine may be 30,000 to 50,000 yuan, which is relatively expensive.
Currently sold overseaspemetinibOriginal drug, Specifications is13.5mg*14 tablets per box may be priced at7about 10,000 yuan (the price may fluctuate due to the exchange rate). The price is relatively expensive. There are also generic drugs produced in other countriespemetiniband their ingredients are the same as those in countryInside、OuterFor sale are basically the same, such as those produced by Laos Pharmaceutical FactorySpecifications4.5mg*14 tabletsThe price of each box may be more than 1,000RMB (the price may fluctuate due to the exchange rate), is still cheaper than the original drug.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















