Detailed Medication Guide for Pemetinib/Pemetinib
1. Common name: pemetinib
Trade name: Pemazyre, Dabotan
Alias: pemetinib, pemetinib,, Pemazyre
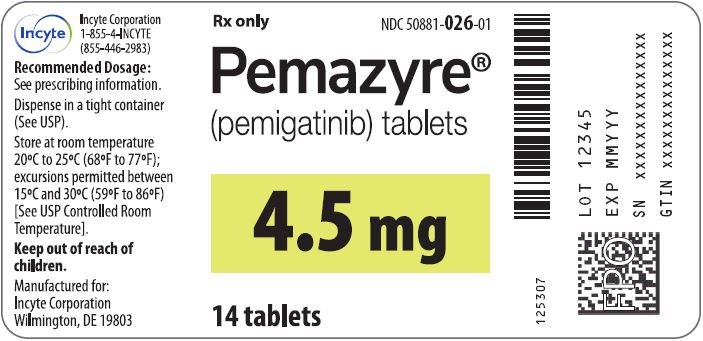
2. Treatment areas
Pemigatinib
1. Cholangiocarcinoma (CCA): Indicated for the treatment of adult patients with locally advanced or metastatic cholangiocarcinoma that remains unresectable after previous treatment. Cholangiocarcinoma in these patients should have fibroblast growth factor receptor 2 (FGFR2) fusions or other rearrangements confirmed by an FDA-approved test.
2. Myeloid/lymphoid neoplasms with FGFR1 rearrangement (MLNs): used to treat adult relapsed or refractory myeloid/lymphoid tumors, and these tumors are accompanied by fibroblast growth factor receptor 1 (FGFR1) rearrangement.
3. Medication Instructions
1. Preparation before use: Use the FDA-approved detection method to confirm the FGFR2 fusion or rearrangement in the patient's cholangiocarcinoma, and then choose to use pemetinib. For patients with relapsed or refractory myeloid/lymphoid neoplasms, there is currently no FDA-approved test to confirm FGFR1 rearrangements for selection of pemetinib therapy.
2. Recommended dosage:
(1) Cholangiocarcinoma (CCA): The recommended dose is 13.5 mg of pemetinib taken orally daily for 14 days, followed by 7 days of discontinuation. Every 21 days constitutes a treatment cycle, and treatment is continued until the condition worsens or intolerable toxicity occurs.
(2) Myeloid/lymphoid neoplasms (MLNs) with FGFR1 rearrangement: The recommended dose is 13.5 mg of pemetinib orally administered daily for continuous administration. Treatment should be continued until disease progression or intolerable toxicity occurs. If the patient misses a dose for more than 4 hours or vomits, he or she should continue to take the next dose as originally planned.
3. Dosage adjustment suggestions:
If patients experience adverse reactions, dose reduction should be considered. Reduce the dose to 9 mg per day for the first time, and further reduce to 4.5 mg per day if it is still intolerable. If the patient cannot tolerate the daily dose of 4.5 mg, pemetinib should be permanently discontinued.
Concomitant use with strong or moderateCYP3A inhibitors should be avoided. If this cannot be avoided, the pemetinib dose should be reduced from 13.5 mg to 9 mg or less. After discontinuation of a CYP3A inhibitor, pemetinib can be resumed at the original dose after 3 plasma half-lives. For patients with severe renal and hepatic impairment, it is recommended that the starting dose of pemetinib is 9 mg, and the dosage schedule should be adjusted according to specific indications.
4. Possible adverse reactions
In clinical trials of cholangiocarcinoma, ≥2% of patients reported serious adverse reactions including abdominal pain, fever, cholangitis, etc. In clinical trials of myeloid/lymphoid tumors with FGFR1 rearrangement, adverse reactions reported by ≥20% of patients included hyperphosphatemia, nail toxicity, etc.
5. Storage conditions
Store pemetinib at room temperature,20°C to 25°C (68°F to 77°F). Allowable temperature fluctuation range is 15°C to 30°C (59°F to 86°F).
6. Drug action mechanism
Pemetinib is a small molecule kinase inhibitor that mainly targetsFGFR1, 2 and 3, with an IC50 value of less than 2nM. By inhibiting abnormal FGFR signaling activation due to FGFR amplification and fusion, pemetinib can reduce cell viability in cancer cell lines, thereby inhibiting tumor cell proliferation and survival. Pemetinib has shown significant antitumor activity in multiple mouse models of human tumors.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















