What is the difference between tenofovir alafenamide fumarate tablets/Veride and whether it is imported or domestically produced?
Tenofovir alafenamide/Veridex (TAF) is a prescription medicine used to treat chronic (persistent) hepatitis B virus (HBV) in adults with stable (compensated) liver disease. Tenofovir alafenamide fumarate tablets may reduce the HBV content in patients, and may also improve the patient's liver condition. Tenofovir alafenamide is a small tablet that is taken once a day with food. Patients should not stop taking this medicine unless told by their doctor; this is because HBV infection in the body may worsen (sudden attack) if the medicine is stopped suddenly. A "flare" is when the HBV virus infection suddenly returns in a worse way than before.
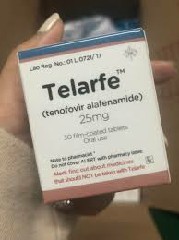
Tenofovir alafenamide fumarate tabletsThe original drug has been launched in China and has been included in the medical insurance. It is sold under the name of tenofovir alafenamide fumarate tablets (Vemlidy/Vemlidy). The original drug is produced by the American pharmaceutical company Gilead Sciences. Most of the common specifications are 25mg*30 tablets. Thereforetenofovir alafenamide fumarate tabletsthe original drug is an imported drug. Imported drugs generally follow more stringent production standards and quality controls to ensure the quality and safety of the drugs. Gilead Sciences will strictly follow relevant regulations and standards during the production process.
Domestic tenofovir alafenamide tablets also exist. There are many pharmaceutical companies in China that produce this drug, such as Qilu Pharmaceutical, North China Pharmaceutical, Chengdu Yuandong Biopharmaceutical, etc. The products of these companies have been certified by the State Food and Drug Administration and are available to patients on the market, and have basically the same pharmaceutical ingredients as imported drugs. Therefore, for patients, the emergence of domestically produced tenofovir alafenamide tablets not only increases treatment options, but may also be more affordable and reduce treatment costs.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















