Talazoparib (Talzenna) launch status in China in 2024
Talazoparib (Talzenna), this highly anticipated oral poly ADP ribose polymerase (PARP) inhibitor, was carefully developed by Pfizer and has shown significant efficacy in adult patients with HER2-negative locally advanced or metastatic breast cancer who carry harmful or suspected harmful germline breast cancer susceptibility gene (BRCA) mutations. Not only that, it also shows great potential in the treatment of metastatic castration-resistant prostate cancer (mCRPC) with homologous recombination repair (HRR) gene mutations.
However, as of2024, talazoparib has not yet been officially launched in China. This news is undoubtedly a pity for patients who are looking forward to this new drug. But even in the face of such difficulties, patients can still obtain talazoparib by purchasing it overseas.
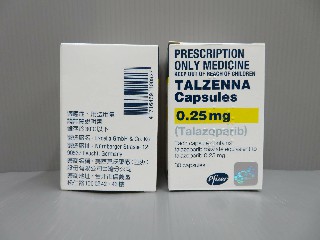
The original drug of talazoparib produced by Pfizer has a higher price on the market. The price of medicines with the specification of 0.25mg*30 pills is about 18,000 yuan, while the price of medicines with the specification of 1mg*30 pills is close to 50,000 yuan. Such a price is undoubtedly a heavy burden for patients with limited financial conditions.
Fortunately, in overseas markets, patients can also choose to purchase Lao element version of generic drugs. The specification of this generic drug is 1mg*30 pills per box, and the price is about 4,000 yuan, which is more affordable than the original drug. Of course, patients must choose formal channels when purchasing to ensure the quality and safety of drugs.
In order to ensure that the medicines purchased are authentic and reliable, patients can make judgments by checking the packaging, labels, and anti-counterfeiting marks of the medicines when purchasing. Genuine drugs usually have clear packaging and labels, and are equipped with anti-counterfeiting marks. Patients can verify the authenticity of the drugs by scanning QR codes and other methods.
In short, although talazoparib will not be available in China until 2024, patients can still obtain it by purchasing it overseas. When purchasing, patients should choose formal channels, pay attention to distinguishing the authenticity of drugs, and ensure their own medication safety.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















