What is Opsynvi (Macitentan and Tadalafil)?
Opsynvi (Macitentan and Tadalafil) is an endothelin receptor antagonist (ERA) and phosphodiesterase 5 (PDE5) inhibitor combination used to treat pulmonary arterial hypertension (PAH). Opsynvi consists of a combination of two previously approved drugs. Macitentan was initially approved under the trade name Opsumit, and Tadalafil was initially approved under the trade name Adcirca as monotherapy for the treatment of pulmonary arterial hypertension (PAH, WHO Group I).
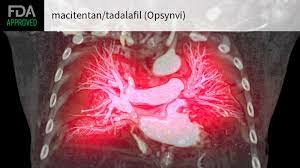
Opsynvi is mainly suitable for the chronic treatment of pulmonary arterial hypertension (PAH, WHO group I) in adult patients with World Health Organization functional class (FC) II-III. Pulmonary hypertension is a rare, progressive, and life-threatening vascular disease characterized by constriction of small pulmonary arteries and increased blood pressure in the pulmonary circulation, ultimately leading to right heart failure. Macitentan in Opsynvi works to reduce the risk of clinical deterioration events and hospitalization, while Tadalafil works to improve exercise capacity.
The U.S. Food and Drug Administration (FDA)approval ofOpsynvi is based on results from the pivotal Phase 3 DUE study, in which Opsynvi showed greater reductions in pulmonary vascular resistance (PVR) after 16 weeks than Macitentan or Tadalafil monotherapy. Opsynvi is available as tablets that can be taken by mouth once daily at a dose provided by your doctor, with or without food.
Due to the risk of embryo-fetotoxicity, Opsynvi products carry a boxed warning on the label. For all female patients, Opsynvi is only available through a restricted program called the Risk Evaluation and Mitigation Strategy (REMS) for Macitentan-Containing Products.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















