The original manufacturer and drug description of Dabotan pemetinib
Dabotan, also known as pemetinib, is a targeted therapy for patients with certain types of cholangiocarcinoma. The drug has been approved by the Food and Drug Administration for marketing in China and has attracted the attention of many patients. The following is a brief description of the original manufacturer of Dabotan and the relevant description of the drug.
1. Original research manufacturer
Dabotan (pemetinib) was developed by the American companyIncyte, and its English trade name is Pemazyre. Incyte is a biotechnology company focused on the discovery, development and commercialization of innovative drugs to address patients with serious unmet medical needs. The development of pemetinib reflects Incyte's deep R&D strength and spirit of continuous innovation in the field of tumor treatment.
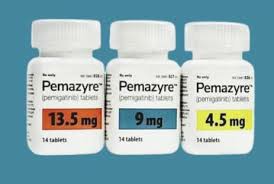
2. Drug description
1. Indications: Dabotan (pemetinib) is approved for the treatment of adult patients with advanced, metastatic, or unresectable cholangiocarcinoma who have received at least one prior systemic therapy and have been confirmed to have fibroblast growth factor receptor2 (FGFR2) fusion or rearrangement. The establishment of this indication provides new treatment options for these patients. Before use, please consult your doctor to ensure it is suitable for your type of disease.
2. Specifications and prices: Dabotan comes in various specifications, the most common of which are 4.5mg and 9mg tablets. In terms of price, prices may vary depending on specifications and regions. Taking the specification of 4.5mg*14 tablets as an example, the retail price is about 20,000 to 30,000 yuan/box.
3. Mechanism of action: Pemetinib is a selectiveFGFR receptor tyrosine kinase inhibitor. It mainly inhibits the abnormal activation of FGFR signaling, thereby inhibiting the proliferation, survival, migration and angiogenesis of tumor cells, thereby achieving the purpose of treating malignant tumors.
4. Medical insurance situation: At present, dabotan (pemetinib) has not been included in my country’s medical insurance catalog, so it cannot be reimbursed through medical insurance.
In summary, Dabotan (pemetinib) is the drug approved by the U.S.Innovative drugs developed by Incyte provide new treatment options for patients with specific types of cholangiocarcinoma. Although the price is higher and it is not yet covered by medical insurance, some patients with limited economic conditions can still enjoy the therapeutic benefits of this innovative drug by purchasing generic drugs.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















