Instructions for Rytelo (Imetelstat)
1. Name:Rytelo (Imetelstat)
2. Indications:
Rytelo (Imetelstat) is indicated for the treatment of adult patients with low-to-moderate risk myelodysplastic syndrome (MDS) who have transfusion-dependent anemia, require 4 or more red blood cell units for more than 8 weeks and are unresponsive or ineligible for an erythropoiesis-stimulating agent (ESA).
3. Usage and dosage:
1. Recommended dose: The recommended dose of Rytelo is 7.1mg/kg, intravenously infused for 2 hours every 4 weeks. RYTELO should be discontinued if the patient does not experience a reduction in red blood cell (RBC) transfusions after 24 weeks of treatment (6 doses) or if unacceptable toxicity occurs at any time.
2. Recommended premedication: At least30 minutes before administration. Patients may need to take the following pretreatment medications to prevent or reduce potential infusion-related reactions: Diphenhydramine (or equivalent) 25 mg to 50mg intravenously or orally; hydrocortisone (or equivalent) 100mg to 200mg intravenously or orally; monitor patients for adverse reactions for at least one hour after completion of infusion.
3. Dose adjustment: Patients may experience intolerable adverse reactions after using Rytelo. The doctor will temporarily delay the dose, reduce the dose or stop treatment according to the severity of the condition. The initial dose can be reduced to once every 4 weeks 5.6 mg/kg; the second dose may be reduced to 4.4 mg/kg every 4 weeks; if the patient cannot tolerate the lowest dose level of 4.4 mg/kg, Rytelo treatment should be permanently discontinued.
4. Adverse reactions:
In clinical studies of Rytelo, the most common adverse reactions (≥10%) were fatigue, arthralgia/myalgia, COVID-19 infection, and headache; laboratory abnormalities included thrombocytopenia, leukopenia, neutropenia, increased aspartate aminotransferase (AST), increased alkaline phosphatase, increased alanine aminotransferase (ALT), and prolonged partial thromboplastin time.
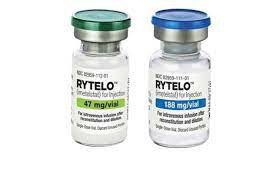
5. Supply and storage:
1. InjectionRytelo is a preservative-free white to off-white or yellowish lyophilized powder available in 47 mg single-dose vials and 188 mg single-dose vials in the original carton refrigerated at 2°C to 8°C (36°F to 46°F). Do not freeze.
2. If the diluted Rytelo solution is not used immediately, store it at room temperature 20°C to 25°C (68°F to 77°F); from reconstitution of Rytelo to completion of intravenous administration The total time of infusion should not exceed 18 hours; when stored refrigerated at 2°C to 8°C (36°F to 46°F), nor should it exceed 48 hours after reconstitution.
6. Special groups:
Based on animal studies,Rytelo may impair fertility in female animals of reproductive potential, and the effects on fertility are reversible. Therefore, it is recommended that females of reproductive potential use effective contraception during treatment with Rytelo and within 1 week after the last dose; lactating women should not breastfeed during treatment with Rytelo and within 1 week after the last dose.
7. Mechanism of action:
The ingredient in Rytelo, Imetelstat, is an oligonucleotide human telomerase inhibitor that binds to the template region of the human telomerase (hTR) RNA component, inhibiting telomerase enzymatic activity and preventing telomere binding. Increased telomerase activity and human telomerase reverse transcriptase (hTERT) RNA expression has been reported in myelodysplastic syndromes and malignant stem and progenitor cells. Non-clinical studies have shown that treatment with Imetelstat results in reduced telomere length, reduced proliferation of malignant stem and progenitor cells, and induction of apoptotic cell death.
8. Non-clinical toxicology
Carcinogenicity studies have not been conducted withImetelstat. In vitro, Imetelstat is non-mutagenic in the bacterial mutagenicity test (Ames test) and non-clastogenic in the chromosomal aberration test using cultured human peripheral blood lymphocytes. In an in vivo mouse micronucleus test, Imetelstat was not genotoxic at intravenous doses up to approximately 10 mg/kg. Fertility studies have not been conducted with Imetelstat. In a general toxicology study, female monkeys administered 14.1 mg/kg once weekly for 9 months showed endometrial atrophy. This effect was observed at a mean exposure (based on AUC) of approximately 14.4 times the human exposure at recommended clinical doses. This finding was not seen in animals after the 14-week recovery period.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















