What is deuterated coxitinib?
Deucravacitinib is an oral, highly selective allosteric inhibitor of tyrosine kinase 2 (TYK2), developed by Bristol-Myers Squibb (BMS). In recent years, it has received widespread attention globally, especially in the treatment of psoriasis.
Deuterated colexitinib is mainly used to treat adult patients with moderate to severe plaque psoriasis who are suitable for systemic therapy or phototherapy. The drug has shown significant efficacy in multiple clinical studies. For example, in a pivotal clinical trial, up to 68.8% of patients treated with deuterated colexitinib achieved PASI after 16 weeks. 75 (that is, the skin lesions are improved by 75% or more). This data is significantly higher than the placebo group, proving its excellent therapeutic effect.
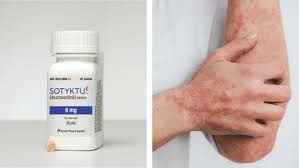
Deuterated colexitinib inhibits the activity of TYK2, thereby blocking the signaling of multiple cytokines involved in the pathogenesis of psoriasis, such as interleukin(IL)-23, IL-12 and I type interferon(IFN). This innovative mechanism of action makes deuterated colexitinib outstanding in the treatment of psoriasis.
In terms of safety, deuterated colexitinib also showed good performance. Although some patients may experience some adverse reactions during use, most are mild and controllable. Common adverse reactions include upper respiratory tract infection, elevated blood creatine kinase, herpes simplex, etc., but the incidence of serious adverse reactions is relatively low.
Deuterated colexitinib is the world's first approved TYK2 inhibitor, and its unique efficacy and safety have been widely recognized. In 2022 in September , the drug was approved for marketing in the United States, and has since gradually gained recognition and application worldwide. This not only provides new treatment options for psoriasis patients, but also marks a major advancement in the field of psoriasis treatment.
Although deuterated colexitinib has shown significant efficacy and good safety in the treatment of psoriasis, there are still some matters that need to be paid attention to during use. For example, this drug should be contraindicated in patients with known hypersensitivity to deuterated colexitinib or any component of the formulation. In addition, patients should be monitored regularly during use to ensure the safety and effectiveness of the drug.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















