Medicare price of fenelidone
Finerenone (Finerenone), as an innovative original drug, successfully passed the strict approval of the National Medical Products Administration in June 2022 and was officially launched in China. The drug, sold as fenelidone tablets under the trade name Kerendia, has unique pharmacological effects that provide a new treatment option for adult patients with chronic kidney disease associated with type 2 diabetes.
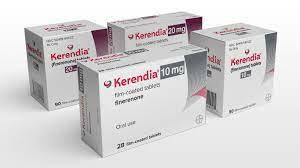
Finelidone, a drug marketed overseas under the trade name"Kerendia", acts as a mineralocorticoid receptor antagonist that works by blocking the action of certain endogenous steroids that may cause damage to the heart and kidney systems. Adults with type 2 diabetes who also have chronic kidney disease, which worsens over time and leads to kidney failure, can be treated with fenelidone. This treatment can reduce the chance of worsening kidney disease, heart failure requiring hospitalization, life-threatening vascular disease, or heart attack.
The common specifications of fenelidone tablets on the domestic market are10mg*14 tablets and 20mg*7 tablets. The price of each box may be several hundred yuan. Currently, it has passed the relevant regulations of the National Medical Insurance Bureau and has officially entered the ranks of Class B medical insurance reimbursement. The implementation of this policy is undoubtedly a huge boon to the majority of patients who are eligible for fenelidone. Under relevant policies, medical insurance can be used for reimbursement, thereby reducing the financial burden and allowing better access to treatment. However, due to different reimbursement ratios in different regions, the price after reimbursement may be different. For specific relevant information, please consult the local medical insurance bureau or hospital.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















