Is Sotolaxib (AMG510) a tablet?
Sotorasib (AMG510, Sotorasib) is an oral tablet that is a targeted drug that is often used to treat certain types of cancer. AMG510 is a KRAS G12C inhibitor that interferes with the mutated KRAS gene in tumor cells and blocks the activity of the gene, thereby inhibiting the growth and spread of tumor cells. The development of this drug marks an important breakthrough in the treatment of cancers associated with KRAS mutations.
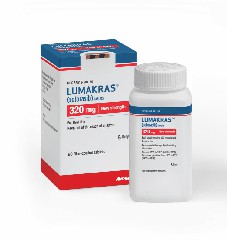
Sotorasiib is a prescription medicine indicated for the treatment of adult patients withKRAS G12C-mutated locally advanced or metastatic non-small cell lung cancer (NSCLC) who have received at least one prior systemic therapy. Sotorasib is available as oral film-coated tablets containing 320 mg, 240 mg or 120 mg of sotorasib, which can be taken orally and conveniently administered by patients at home or in the hospital. Patients should take the medicine according to the doctor's recommendations and dosage instructions to achieve the best therapeutic effect. Taking oral tablets is relatively simple; patients only need to swallow the drug whole or divided according to dosage.
In addition to being easy to take, sotorasiib also has better drug absorption properties as an oral tablet, which can help the drug enter the blood circulation system faster, thereby exerting its efficacy. In addition, the preparation process of oral tablets is relatively mature, and the production, packaging and storage are relatively simple, making it suitable for large-scale production and wide application.
However, when using sotoraxib tablets, patients need to pay attention to taking the drug correctly as directed by their doctor, strictly control the dosage, and be aware of possible side effects and contraindications. At the same time, regular follow-up visits should be carried out to ensure the effectiveness of treatment and physical health. In general, the use of oral tablets makes sotoraxib (AMG510) more convenient and operable in clinical applications, helping to improve patient compliance and treatment effects.
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















