Is Mirikizumab an imported drug?
Mirikizumab (Mirikizumab)-Omvoh is a monoclonal antibody directed against interleukin-23 (IL-23), mainly used to treat autoimmune diseases, such as moderately to severely active ulcerative colitis (UC). IL-23 is a cytokine that plays an important role in immune responses, and its overactivity is closely related to the development of many autoimmune diseases. By inhibiting the activity of IL-23, militizumab can help regulate the immune system response, thereby reducing inflammation and improving patients' clinical symptoms.
Milizumab is usually administered by intravenous infusion and subcutaneous injection. During the induction phase, patients need to receive a dose of 300mg, which will be intravenously infused at weeks 0, 4 and 8 to quickly control the condition. During the maintenance phase, the recommended dose is 200 mg, with patients receiving an initial subcutaneous injection at week 12 and then every 4 weeks. This mode of administration is designed to achieve rapid relief and maintain effects during subsequent treatments.
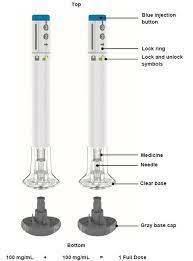
Regarding the origin of this drug, Milizumab was developed by Eli Lilly and Company of the United States. If the drug can be marketed in China, it will be an imported drug. As the global demand for innovative drugs continues to increase, militizumab, a new generation biopharmaceutical product, has been approved for marketing in many countries and regions. After being imported into the domestic market, it aims to provide more treatment options for patients in China and other countries, especially those who are no longer effective or intolerant to traditional treatments.
It is worth noting that although militizumab has shown good results in treatment, you still need to pay attention to potential side effects when using this drug, including the risk of infection, allergic reactions, etc. Therefore, before starting treatment, patients should fully communicate with their doctors to understand the drug's indications, possible side effects, and monitoring options.
Reference materials:https://pubmed.ncbi.nlm.nih.gov/37465925/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















