What is the price list for Zotuximab/Veloximab?
Zolbetuximab - Vyloy) is the world's first monoclonal antibody drug targeting Claudin 18.2 (CLDN18.2), a new target, which is a milestone in the field of gastric cancer treatment. According to international and domestic approval information, the main indication of the drug is the treatment of locally advanced unresectable or metastatic gastric cancer and gastroesophageal junction adenocarcinoma, provided that the positive expression of CLDN18.2 is detected in the patient's tumor tissue. This indication setting makes it the only CLDN18.2-targeted drug currently entering clinical practice, providing a new precision treatment plan for gastric cancer patients.
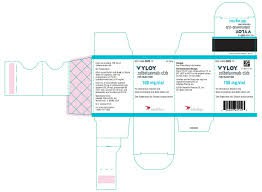
In China,zolbetuximab has been officially launched under the trade name "Veloyi", which means that patients no longer need to rely on cross-border drug purchases to obtain the drug in China. However, it should be pointed out that this drug has not yet been included in the national medical insurance catalog, and the financial burden that patients need to bear during treatment is still heavy. According to public information, its specification is 100mg/bottle, and the price in the international market is about RMB 7,000 per box. The specific price varies depending on the country, exchange rate and channel. Domestic patients can consult at some large hospitals or designated pharmacies to obtain more accurate price information.
There are currently no generic versions of zotuximab on the market, so all supplies come from originator pharmaceutical companies. This affects the accessibility of drugs to a certain extent and keeps drug prices at a high level. In the future, as the research and development of generic drugs or biosimilar drugs advances, prices are expected to gradually decrease.
In general, the indications of zotuximab/Veloyi are clearly located in Claudin 18.2-positive advanced gastric cancer and gastroesophageal junction adenocarcinoma. As a new breakthrough in precision treatment, it provides new hope for patients.
Reference materials:https://www.astellas.com/en/news/29401
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















